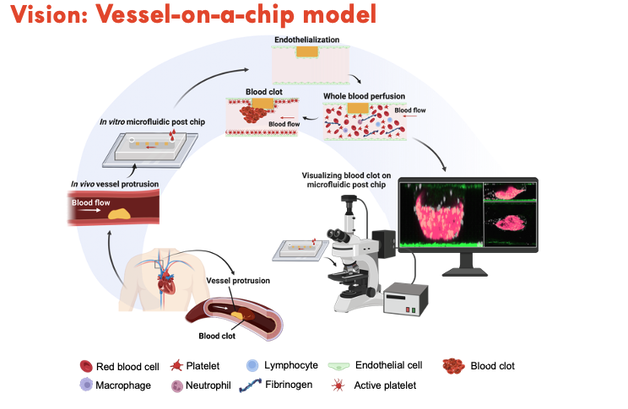
Our ResearchThe ultimate goal of our research is to develop new ‘mechanomedicines’ with ultrasensitive biomechanical and imaging approaches to detect and target disease markers faster, cheaper and more effectively than current technology allows.
|
Developing safer anti-clotting agents derived from “Mother Nature”Therapeutic inhibition of thrombin has been shown to be an effective antithrombotic agent, however, all current inhibitors of this enzyme lead to severe bleeding complications. In an NHMRC joint funded collaboration with Professor Richard Payne at The University of Sydney’s School of Chemistry, we have characterised novel anticlotting agents that have been based around naturally occurring proteins found in saliva of mosquitos.
Our studies have demonstrated that these bug-derived proteins are able to dissolve blood clots in a disease model of thrombosis with fewer bleeding complications. The outcomes of these studies have laid the foundation for the development of safe anticoagulants for the treatment of thromboembolic diseases such as stroke in the future. |
Understanding the mechanisms leading to microvascular dysfunction and poor cerebral perfusion in strokeTargeted therapies have not been effective in improving microvascular perfusion. This is due in part to the lack of suitable animal models and technical difficulties associated with performing real-time imaging on the microvasculature. Using an in vivo model of gut IR injury which allows us access to microvasculature in living animals during IR injury, in combination with precision imaging systems (confocal and scanning electron microscopy), we have observed previously unappreciated in vivo changes within the microvasculature during IR. Our studies demonstrate that ischemia and reperfusion phases trigger phenotypically distinct endothelial dysfunction and death, associated with the onset of distinct vessel occlusion mechanisms involving red blood cells, platelets and neutrophils – ultimately cooperating to lead to MVO in local gut and remote organs. These findings not only demonstrate an intimate spatiotemporal relationship between endothelial injury and vaso-occlusion mechanisms, they also help explain why existing therapies remain ineffective.
|
Biomechanical sensing and blood clot formation:Solving a sticky clotting problem in diabetesThe leading cause of death in diabetes is cardiovascular disease, with up to 70% of deaths relating to the development of blood clots supplying the heart (heart attack) or brain (ischemic stroke). Diabetic individuals are more prone to develop blood clots, and these clots are more resistant to standard anticlotting therapies.
We have discovered a new biomechanical clotting mechanism severely affected by diabetes that is resistant to the beneficial effects of commonly used antithrombotic agents. Studies in the laboratory are also examining the role chronic oxidative stress plays in amplifying blood clotting in diabetes, and the mechanisms by which oxidative stress may modify platelet receptors to enhance adhesion. These studies may identify novel targets with which to treat thrombosis associated with diabetes. |
Developing novel approaches to the treatment of ischaemic strokeThe use of rtPA is associated with significant side effects, limiting its widespread use. We are working on several novel approaches to improve upon existing stroke therapies, making them safer and more effective.
While it is broadly accepted that restoration of blood flow to the brain will limit the progression of cell death and improve patient outcome after stroke, there is evolving evidence that reopening the blocked artery (recanalization) does not always lead to reperfusion of the small vessels of the brain. Critically, failure of cerebral reperfusion correlates with worse prognosis for stroke patients. |
Platelet death as an important regulator of blood clot formationOver the last several years, studies from our laboratory have afforded important insight into the development of platelet procoagulant function, specifically its close resemblance to cell death pathways in nucleated cells. Our studies have demonstrated that procoagulant platelets are dying cells, undergoing a cell death process akin to necrosis, leading to PS exposure and thrombin generation. We have also found a novel role for an adaptor protein – 14-3-3z – in regulating platelet death necessary for blood clot growth and stability. We aim to determine whether therapeutic targeting of these pathways represents a safe and effective way of reducing thrombin generation in vivo without increasing bleeding risk.
|
Developing a novel preclinical ischaemic stroke modelOur ongoing studies have successfully developed a novel in vivo model of thrombolysis (iCAT) that for the first time allows us the ability to examine the efficacy of novel drugs including novel thrombin inhibitors to facilitate clot lysis (recanalization), restore perfusion in the brain, as well as determine whether cerebral damage and cognitive impairment associated with stroke are reduced. We are using this model to assess the efficacy of currently approved and novel anticlotting agents to facilitate stroke treatment.
|